A paper entitled: “Neurological complications after first dose of Covid-19 vaccines and SARS-CoV2-infection” has found that[1]–
“Emerging reports of rare neurological complications associated with COVID-19 infection and vaccinations are leading to regulatory clinical and public health concerns. We undertook a self-controlled case series study to investigate hospital admissions from neurological complications in the 28 days after a first dose of ChAdOx1nCoV-19 (n= 20 417 752) or BNT162b2 (n= 12 134 782) and after a SARS-CoV-2-positive test (n= 2 005 280). There was an increased risk of Guillain–Barré syndrome (incidence rate ratio (IRR) 2.90; 95% confidence interval (CI): 2.15–3.92 at 15–21 days after vaccination) and Bell’s palsy (IRR 1.29; 95% CI: 1.08–1.56 at 15–21 days) with ChAdOx1nCoV-19.
There was an increased risk of hemorrhagic stroke(IRR 1.38; 95% CI: 1.12–1.71 at 15–21 days) with BNT162b2.”
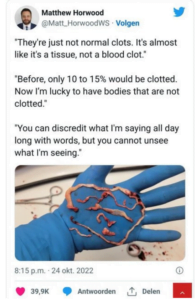
Another study entitled: “Effect of COVID-19 mRNA vaccine on in vitro glial cells of the brain studied by Raman spectroscopy and imaging”[2]depicts how researchers have studied how the Covid-19 vaccines altered the biochemical composition of glial and glioma brain cells in vitro. COVID-19 mRNA has been recovered from vaccinated cerebrospinal fluid indicating that it can cross the blood-brain barrier (BBB). Furthermore even without crossing the BBB several cytokines induced by COVID-19 infection cross the BBB and affect CNS function. They also found that when the mRNA reaches the brain it infects astrocytes and triggers neuropathological changes that contribute to the structural and functional alterations in the brain. “The researchers have also raised concerns that the lipid nanoparticles (LNPs) can diffuse quickly to the CNS through the olfactory bulb or blood.” Raman spectroscopy was used to examine symptoms like impaired “consciousness and vision nerve pain dizziness nausea and vomiting seizures hemiplegia stroke ataxia and cerebral hemorrhage in human brain glial and glioma cellsin vitro.”
Another study entitled: “Thromboembolic events and haemorrhagicstroke after mRNA (BNT162b2) and inactivated (CoronaVac) covid-19 vaccination: A self-controlled case series study”[3]had the following findings:
“A total of 5 526 547 doses of BNT162b2 and 3 146 741 doses of CoronaVac were administered. A total of 334 and 402 thromboembolic events and 57 and 49 haemorrhagicstroke cases occurred within 28 days after BNT162b2 and CoronaVac vaccination respectively. The crude incidence of thromboembolic events and haemorrhagic stroke per 100 000 doses administered for both covid-19 vaccines were smaller than that per 100 000 SARS-CoV-2 test positive cases. The modified SCCS detected an increased risk of haemorrhagicstroke in BNT162b2 14-27 days after first dose with adjusted IRR of 2.53 (95% CI 1.48-4.34) and 0-13 days after second dose with adjusted IRR 2.69 (95% CI 1.54-4.69).”
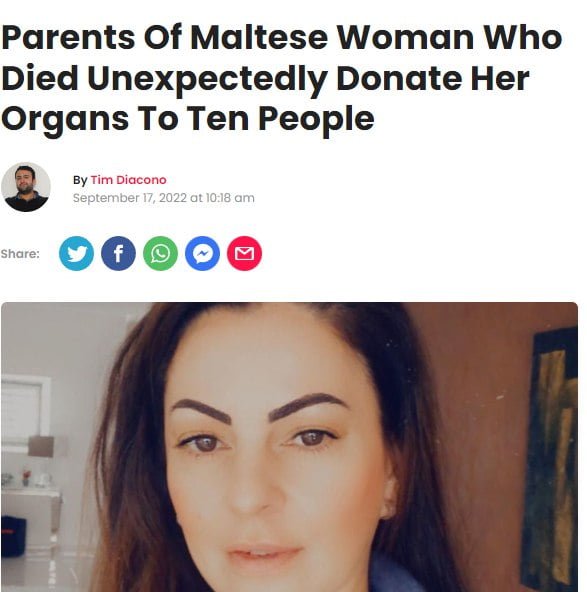
And then we have Lovin Malta’s article about the unfortunate and saddening sudden death of a young Maltese woman who “unexpectedly died of a brain haemorrhage.”[4]
Can Lovin Malta do some more investigation?
[1]https://www.nature.com/articles/s41591-021-01556-7
[2]https://www.news-medical.net/news/20220308/Effect-of-COVID-19-mRNA-vaccine-on-in-vitro-glial-cells-of-the-brain-studied-by-Raman-spectroscopy-and-imaging.aspx
[3]https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370(22)00234-6/fulltext
[4]https://lovinmalta.com/news/news-human-interest/parents-of-maltese-woman-who-died-unexpectedly-donate-her-organs-to-ten-people/