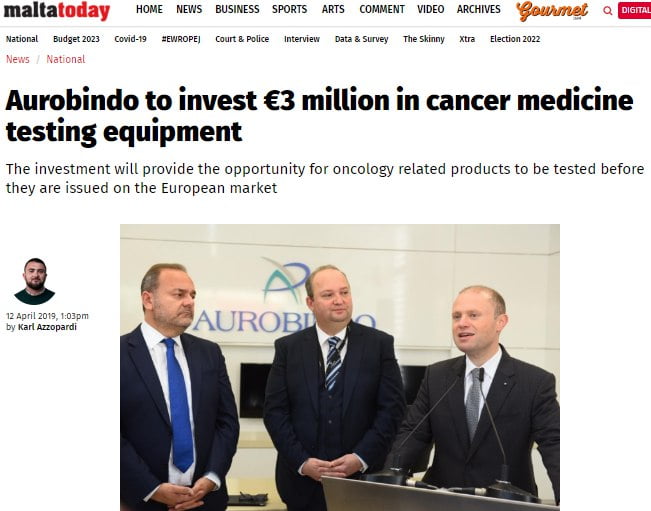
Back in 2019 all local media portals reported that Aurobindo was investing three million euro in a laboratory for the testing of cancer medicines. The said Indian pharmaceutical company based in Hal Far had set up operations in Malta in 2008 and such investment was announced on its 10thyear anniversary here.
With such a new laboratory the company was set up to go for oncology testing in Malta with its facilities already being the company’s first European GMP-certified facility. It is also one of the largest generic drug manufacturers in the world and the second largest generics producer for the U.S.
By 2019 Aurobindo Pharma was exporting to over 125 countries with annual turnover in excess of $900 million. Its undertaking was supported by Malta Enterprise and the Malta Medicines Authority.
Could it also be the case that they will be getting vaccinated patients with Covid-19 jabs induced cancer over here for treatment? We already had the filthy corrupt to the bones messy organisation the WHO which is no more credible than the EU and the WEF hailing the new system of cancer care which is being offered at the Sir Anthony Mamo Oncology Centre at Mater Dei Hospital[1]. There is no doubt that the doctors nurses and staff are doing an excellent job. The WHO has commended this system as “innovative” and has advised other countries to consider following in Malta’s footsteps. But with a growing movement of lack of credibility towards the WHO especially in the way it has “managed” Covid-19 then its praise and statement might lead for more questions.
Did Dr Fearne pass a good word to the WHO by any chance since the international body he is in does give advice to organisations like the same WHO?
But there is more to Aurobindo which the government is not telling you. Firstly the company’s manufacturing plant in India had run into issues with the US Food and Drug Association. These issues revolve around regulatory problems and problems in procedures that are supposed to prevent microbiological contamination of sterile products. Such findings had come as it was expanding its capabilities in the US and Europe[2].
Secondly it has been repeatedly knocked by the same FDA “for its role in global “sartan” recalls for allowing tainted active pharmaceutical ingredients (APIs) into its drugs”. Aurolife a subsidiary of Aurobindo “ended up recalling 2 352 bottles with 1000 tablets each of generic statins after certain lots were found to have incorrect or missing labels and sometimes missing expiration dates”[3].
Thirdly it so happened that back in 2009 Pfizer Inc PFE.N “agreed to license an array of generic pills and injectable medicines from Aurobindo.”[4]
Fourthly it so happened that two years later in 2011 Pfizer Inc has entered into other series of licensing agreements with Aurobindo Pharma Ltd. “to commercialize medicines that have come off patent and have lost market exclusivity in the US and Europe.”[5]
Lastly Aurobindo Pharma has signed a non-exclusive license agreement with Medicine Patent Pool (MPP) to manufacture and commercialise Pfizer’s famous Covid-19 oral pill Paxlovid under the name of Nirmatrelvir. I remind the public that this Paxlovid has received emergency use authorisation by the same FDA and the UK Medicines and Healthcare products Regulatory Agency (MHRA) for the treatment of Covid-19 in patients at high-risk of progressing to serious illness[6].
Because after their “safe and effective” Covid-19 vaccines they are selling you Paxlovid while Aurobindo whose oncology testing laboratory is based in our little Malta will sell you Nirmatrelvir.
[1]https://timesofmalta.com/articles/view/hails-new-system-cancer-care-malta.991436
[2]https://www.cleanroomtechnology.com/news/article_page/FDA_issues_concerns_for_Aurobindo_manufacturing_plant/140560
[3]https://www.fiercepharma.com/manufacturing/aurobindo-recalls-mislabeled-statin-tablets-off-u-s-market
[4]https://www.reuters.com/article/pfizer-aurobindo-idINN0240167220090303
[5]https://www.uspharmacist.com/article/pfizer-and-aurobindo-sign-licensing-agreement
[6]https://economictimes.indiatimes.com/industry/healthcare/biotech/pharmaceuticals/torrent-aurobindo-receive-licenses-to-sell-pfizers-covid-pill-paxlovid-in-india-low-income-countries/articleshow/90297321.cms?from=mdr